Fair Use Is A Use Permitted By Copyright Statute That Might Otherwise Be Infringing. They reactivity depends on the type of alkyl halide (F, Cl, I, Br), its substitution (primary, secondary, tertiary) and the desired reaction (SN1, SN2, E1, E2). 
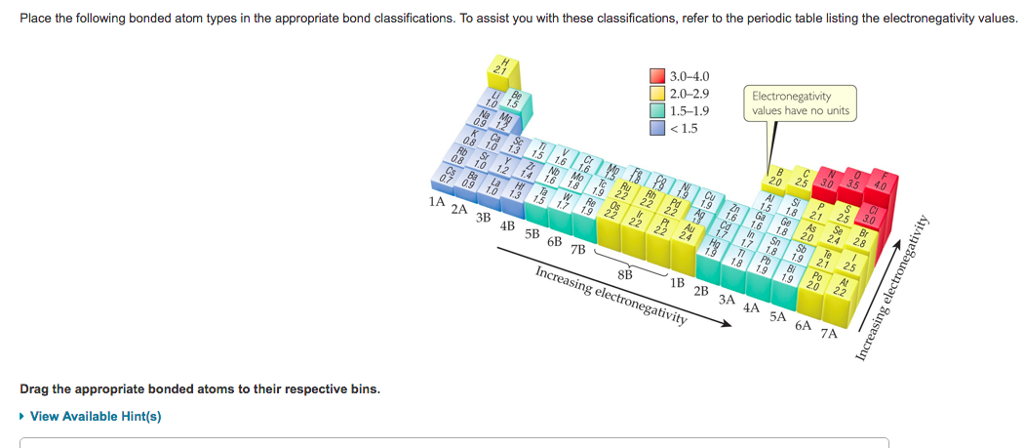
some other website seem to say that, which one is correct? In the example below, the difference between cis and trans alkenes is readily apparent. 4-(Carboxymethylene)-2,5-heptadienedioic acid. Hence determining the priority order is a key step in naming of the organic compounds. If Halogens have higher perioity than Nitro why the Nitro group is written after the Bromine. In the case where a nitrogen has four bonds to carbon (which is somewhat unusual in biomolecules), it is called a quaternary ammonium ion. *Examples does not contradict the ALPHABETICAL rule. a) a compound with molecular formula C6H11NO that includes alkene, secondary amine, and primary alcohol functional groups. Respected sir, I have a doubt which may be small for you, but it brings more confusion if sulphonic acid and carboxylic acid are given which should be given priority first sir. identify the functional groups present in an organic compound, given its structure. Sponsored by PureCare Knee Protector The male sex hormone testosterone contains ketone, alkene, and secondary alcohol groups, while acetylsalicylic acid (aspirin) contains aromatic, carboxylic acid, and ester groups. Amines are characterized by nitrogen atoms with single bonds to hydrogen and carbon. The sum of the locants is 1 + 4=5 from both the directions. On the table, alkene comes before alkyne but while naming molecule with both alkene and alkyne, alkyne will be the suffix. When an alkeneand an alkyne are present in a molecule, which takes priority? What's The Alpha Carbon In Carbonyl Compounds? -CHO 8. Its an arbitrary agreement by IUPAC, although note that there is some correlation between the oxidation state of the carbon and the priority (more oxidized groups tend to be higher priority). Chloroform is a useful solvent in the laboratory, and was one of the earlier anesthetic drugs used in surgery. We will learn more about the structure and reactions of aromatic groups in Chapter 15. Among the amine derived functional groups, the priority is given as follows. 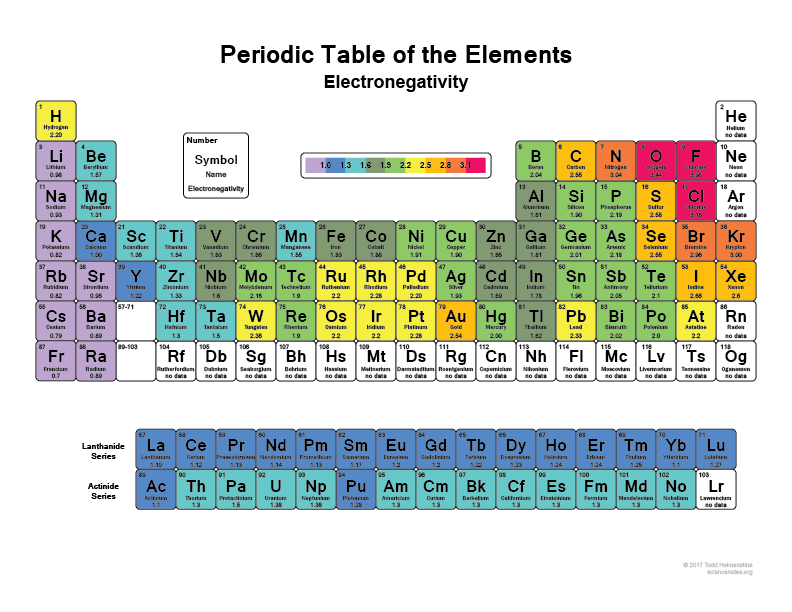
Systematic screening revealed that an epoxide functionality possesses the special combination of stability and reactivity which renders it stable toward proteins in solution but reactive on the protein surface outside the active site (proximity-induced reactivity). In this paper, biochar (BC) was prepared from discarded grapefruit peel and modified to prepare magnetic biochar (MBC). Molecules with carbon-nitrogen double bonds are called imines, or Schiff bases. 
where does benzene stand in priority table? This is wonderful!, ,, ,, ,, , Chemical Compound Suffixes - Chemical Literature,, Best 17 Alkene Group List - Edu Learn Tip. Ammonia is a special case with no carbon atoms. Depending on whether or not the halogen substituent is above or below the alkoxy alphabetically is the key thing. 4,4-Diphenylmethane bismaleimide (BMI)/2,2-diallylbisphenol A (DABPA) resin was modified with a multifunctional thiol containing isocyanuric ring and long-chain aliphatic unit (tris-isocyanurate (TEMPIC)). For simplification, we have included widely used functional groups here. Functional Groups and Reactivity Functional groups play a significant role in directing and controlling organic reactions. Because of this, the discussion of organic reactions is often organized according to functional groups. Next preference is given to sulfonic acids. Impaired cerebrovascular reserve has been suspected to explain hemodynamic stroke. In the case of halogens and ethers its the alkane which has highest priority (the suffix) and the halogens / alkoxy groups are prefixes that will be ranked based on alphabetical order. The priority order of functional groups in IUPAC nomenclature is based on a relative scale where all functional groups are arranged in the decreasing order of preference. The structure of capsaicin, the compound discussed in the beginning of this chapter, incorporates several functional groups, labeled in the figure below and explained throughout this section.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
